Electroplating is a branch of science called Electrochemistry, which studies the deposition of electrolytes on the surface of metals. The process of applying a metal coating to any product is also called. For protection against corrosion, for example, galvanizing or chromium plating is used.
Despite the fact that the process of applying electrolytes is quite complicated and unsafe, decorative, that is, intended for decorating and protecting electroplating at home, is becoming popular today.
There is another interpretation of this term, but more on that later.
General idea of electroplating
Electroplated coating can be both technological and decorative-protective. It is a thin surface layer of metal with a good aesthetic appearance (gold, silver) or anticorrosion properties (zinc, copper) on metal or plastic products.
In general, metal plating at home looks simple.  Anodes are lowered into the bath with electrolyte, which are connected to the "plus", and between them - as a cathode - the workpiece connected to the "minus". When the electric circuit closes, the metal that is contained in the electrolyte is deposited on the cathode, that is, the product.
Anodes are lowered into the bath with electrolyte, which are connected to the "plus", and between them - as a cathode - the workpiece connected to the "minus". When the electric circuit closes, the metal that is contained in the electrolyte is deposited on the cathode, that is, the product.
Electroplating Equipment
At home, and equipment you can do it yourself. First of all, you need a power source, since the process proceeds under the influence of an electric current. In terms of current strength, home masters diverge, calling them in a very large range. But a prerequisite is the presence of a voltage regulator for a smooth change in output power. The current must be constant, therefore, a rectifier of its own or factory manufacture is called as a source.
Craftsmen fit a welding machine for electroplating at home.
An electrolyte needs a container (bath) made of chemically neutral material. This can be a glass or plastic vessel of sufficient size to fit in the part for processing and the required amount of electrolyte. It must withstand high, up to 80 ° C, temperature and be strong enough.
Still need anodes, the area of which should be greater than the area of the part. They serve to supply electric current to the electrolyte and its uniform distribution over the part. In addition, they must compensate for the loss of metal in the electrolyte, which is released during the coating of products, and perform some oxidative processes.
It is better to use heating devices to bring the electrolyte to the desired temperature with the ability to regulate thermal conditions. For example, a domestic gas stove is not suitable for this purpose. Usually they use a small electric stove and iron, in which you can set the required temperature of the sole.
Electrolyte Preparation Equipment
To store chemicals and electrolytes you need glassware with ground lids.
For the preparation of electrolytes, the necessary substances must be measured with an accuracy of one gram. For this, appropriate scales are needed. They can be purchased or made independently, using Soviet coins as weights, the nominal value of which exactly corresponds to the weight.
Electroplating at home is also the preparation of an electrolyte from chemicals obtained in a difficult way.Specialized companies selling such high-quality substances are not uncommon, but their activities are controlled, and working with them requires special permits, even for legal entities. Hazardous chemicals are not sold to individuals.
Preparation of the product for electroplating
Having collected everything you need (having found in the garage, having purchased and installed with your own hands), you can begin to prepare the part. This stage of work is very responsible. Successful electroplating at home requires thorough cleaning of the part. The quality of the plating also depends on the surface preparation. Therefore, depending on the desired result, the part is not only degreased. In many cases, sandblasting is required, followed by sanding with sandpaper and sanding pastes.

For degreasing, parts are washed either in pure organic solvents, acetone, alcohol or gasoline, or in prepared solutions. For example, for degreasing steel or cast iron parts, hot, up to 90 ° C, solutions with caustic soda, soda ash, water glass, sodium phosphate are used.
Non-ferrous metal products are processed in solutions with laundry soap and the same sodium phosphate.
The oxide film is removed in special decapsulating solutions with hydrochloric or sulfuric acid.
Work Safety
Electroplating at home (chromium plating, copper plating, nickel plating) is a dangerous process. Firstly, due to the toxicity of the substances used. Secondly, because of the need to heat solutions to high temperatures. And thirdly, because of the risk associated with any electrochemical processes.
Do not take the term "at home" literally. It is better to do electroplating in a garage or other non-residential premises with good ventilation. The electrical equipment used must be earthed.
And it is very important to take care of personal safety. In no case should you drink or eat anything during the process so as not to swallow harmful vapors at the same time.
You need to work in a respirator, rubber, durable and soft gloves, a tight oilcloth apron and shoes that protect the skin of the legs. It is necessary to protect eyes so that small splashes of reagents do not get into them. It will not be superfluous to re-read special literature on this topic in order to prepare for possible surprises.
Nickel plating of metal products
Nickel plating can be a finish or intermediate (before chrome plating) coating. This process does not cause any special difficulties, the surfaces have a beautiful appearance, and are resistant to weathering.
The electrolyte temperature is not higher than 25 ° C at a current density of up to 1.2 A / sq. dm.
The electrolyte includes aqueous solutions of nickel sulfate, sodium and magnesium, pure sodium chloride (sodium chloride) and boric acid.
The required acidity of the electrolyte - 4-5 pH - is checked by litmus paper. It should turn red. The process takes about half an hour, depending on the current density and thickness of the applied coating.
After the process is complete, the product must be washed in water, dried and polished on a felt circle with polishing paste.
Chrome plating of metal products
Chrome metal parts to increase their hardness and resistance to corrosion, to obtain a decorative effect. Due to the porosity of the chrome coating, copper plating at home or nickel plating, or even applying these coatings in turn, is first performed before it is applied.
A stable chromium plating process is provided by anodes made of an alloy of lead with tin and antimony in the proportions of 85%: 11%: 4%, respectively.
Like electroplating at home, chromium plating is more complex and diverse than nickel plating. Firstly, the shade of the coating depends on the temperature of the electrolyte.At temperatures in the range from 35 to 55 ° С it is brilliant, above 55 ° С - milky, below 35 ° С - matte.
Secondly, the color and physical parameters of the chrome coating depend on the composition of the electrolyte. It can be ordinary protective, dark blue, agate, blue or even black decorative protective. It is difficult to perform a wear-resistant protective coating at home, since high density currents of up to 100 A / sq are needed. dm.
It is difficult to perform a wear-resistant protective coating at home, since high density currents of up to 100 A / sq are needed. dm.
At room temperature and current density up to 5 A / sq. dm it turns out a decorative soft coating.
After the completion of the galvanic process of chromium plating, the part is washed in water, then neutralized in a light solution of ordinary baking soda, again thoroughly washed, dried and polished with special pastes.
Copper plating
Why else might you need electroplating at home? Copper plating is carried out to apply a thin conductive layer with a low resistance to the surface or to protect the base metal of the part.
Putting copper on ferrous metals at home, since cyanide electrolytes are used in this process, is deadly. Therefore, steel or cast iron parts are first nickel-plated, and only then coated with copper in electrolytes from a solution of copper sulfate and concentrated sulfuric acid in room temperature water. The current density is low - up to 2 A / sq. dm.
Aluminum parts also require prior preparation. They are first decapitated (cleaned of the oxide film), then oxidized in an electrolyte from an aqueous solution of sulfuric acid, then washed thoroughly. Only then is electroplating performed at home - copper plating in an electrolyte from copper sulfate and an aqueous solution of sulfuric acid at sufficiently low temperatures, up to 20 ° C, and low current density. As an anode, an ordinary copper plate of a suitable area is used.
Silvering and gilding of products
Galvanic silvering is not only decorative, but also protective and electrically conductive coating with good reflectivity. Products made of ferrous metals are pre-coated with nickel, on which silver is already deposited.
The silver electrolyte consists of silver chloride, potassium iron cyanide, soda ash and distilled water. The working temperature of the electrolyte is not higher than 20 ° C, and high current power is not required - it is not more than 0.1 A / sq. dm. Graphic plates are used as an anode.
The most decorative and therefore seductive electroplating at home is gilding. The process is carried out in hot solutions of gold (up to 5 g per liter of water) with potassium synergistic. In cold solutions, the amount of metal must be tripled. Vapors of synergistic acid are very toxic, therefore, the process of gilding in a hot solution must be carried out only in rooms with a powerful forced extract.
When working with a cold solution with synergistic acid, extreme care must also be taken: do not bend over the bathtub, do not stick your hands into it, especially with fresh abrasions or scratches.
Better yet, replace the poisonous acid with a yellow, bloody salt, that is, iron-ferrous potassium. Before preparing the electrolyte, ordinary gold is converted into bleach using aqua regia, unless, of course, it is possible to purchase the finished one.
How to make electroplating at home? Thoroughly clean the product. Then, if it is made of lead, tin, iron, zinc or steel, it must first be coated with copper. Then, in order to facilitate the pestering of gold, the object must be dipped in a solution of mercuric acid.
The current density should not be higher than 1 A / sq. dm, since at higher currents gold from the electrolyte settles to the bottom of the bath in the form of black powder, and the gilding on the product becomes brownish.
In order to have beautiful gilding, it is necessary to suspend a sufficiently large sheet of gold at the anode.After completion of the galvanic process, the gilded product must be dried in sawdust and polished.
Electroplating at home
Summing up the possibility of applying a galvanic coating at home, we can say that a person who does not understand electrochemistry at least at the initial level should not organize the process - it is too dangerous. In addition to information on various sites and forums, it is necessary to read special literature. In addition, a private person does not have access to reagents. Therefore, if you need to restore chromium plating on one part, it is much cheaper to contact specialists.
Well, if you really want to master the new production yourself, then you also need to ask for help from professionals who will select the necessary safe equipment (after all, production should not be very artisanal), power supplies, electrolytes that do not contain toxic substances, calculate the parameters of the bathtubs and anodes . They will also be taught the processes of applying a metal coating, all those procedures (there are not so many of them) that lead to the desired result.
Electroplating for the face at home
After everything described above, such a name sounds somewhat menacing. In fact, this is just a deep cleansing of the skin with the help of galvanic currents of low power. Under the influence of these currents, skin fats dissolve and come to the surface of the face, covered with a special composition, interacting with which, turn into soap.
Galvanic currents in medicine are used to enhance the work of ions. These are actively charged particles, responsible for many of the working processes in cells. Under the influence of direct current, the ions move, the permeability of cell membranes improves, the production of collagen and elastin fibers starts, increasing the strength, firmness and elasticity of the skin.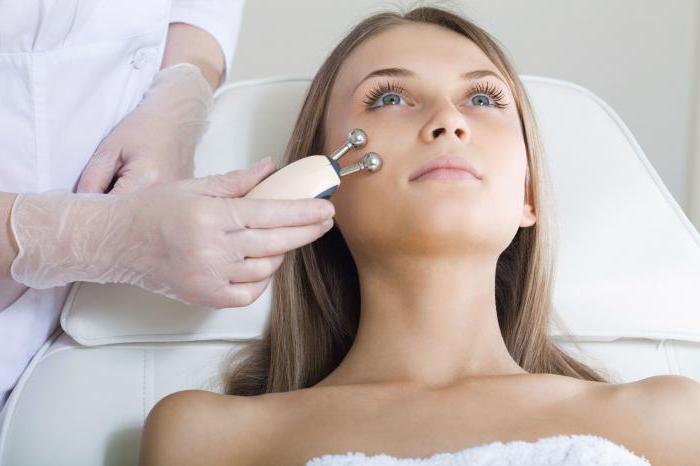
Galvanic currents conduct cosmetics with their own electric charge into the deeper layers of the dermis. That is, the skin healing process takes place from the inside, deep wrinkles disappear, enlarged pores narrow, the skin is intensely saturated with oxygen.
The device “Electroplating + microcurrents” at home M365 Gezatone is by far the most affordable model of a new generation cosmetology device. It helps to get rid of blackheads, smooth wrinkles on aging skin, reduce the manifestations of the vasculature, strengthen blood vessels, improve facial contours, remove the second chin.
Galvanic Facial Cleansing Procedure
Before the procedure begins, the skin is cleaned by conventional means: milk or gel. Then, an alkaline composition is applied. Then begins the electroplating directly at home. Gezatone Beauty Lifting is a special apparatus for the procedure not only of galvanic cleaning, but also for the introduction of nutrients into the skin under the influence of the same currents. The procedure lasts about fifteen minutes - only circular movements with the tip along the massage lines of the face, and nothing more - then the soap formed during the reaction of fats with alkali is washed off with warm water.
But currents are currents, even with the prefix "micro." It is forbidden to undergo a galvanic cleaning procedure for people with an implanted pacemaker, cancer, neurotic conditions, skin diseases.
Is it so effective, according to the manufacturers, electroplating for the face at home, the reviews of women using the devices, can not say with confidence. As a rule, ladies are in a hurry to share their impressions in the first months after the purchase, when the consequences are not yet felt. But still, the reviews are optimistic.
In conclusion, we can say that electroplating at home is not only a dangerous process for applying a metal coating to products that should not be carried out in a living room itself, but also a modern, painless, safe and quite effective procedure for rejuvenating the skin of the face, neck and area neckline.








